You’re staring at a data readout or a complex chemistry textbook, and there it is: Helonium. It sounds like something pulled straight out of a low-budget sci-fi flick or a misplaced element from a Marvel movie. You’ve been told for years that Helium is the “loner” of the periodic table—noble, stable, and completely uninterested in making friends with other atoms. Yet, here you are, seeing a term that suggests Helium has finally decided to bond.
The frustration usually kicks in when you try to find a straight answer. Most sources bury you in calculus or quantum mechanics, leaving you wondering if this is a real substance you can hold or just a theoretical ghost in a lab. If you’ve felt like you’re missing a fundamental piece of the chemical puzzle, you aren’t alone. This isn’t just academic fluff; it’s the story of the first bond ever formed in our universe.
In this guide, we are going to strip away the jargon. We’ll look at exactly how this “impossible” molecule exists, why it’s the strongest acid you’ve never heard of, and why its existence in deep space changes everything we thought we knew about the early cosmos.
What is Helonium? A Plain-English Explanation
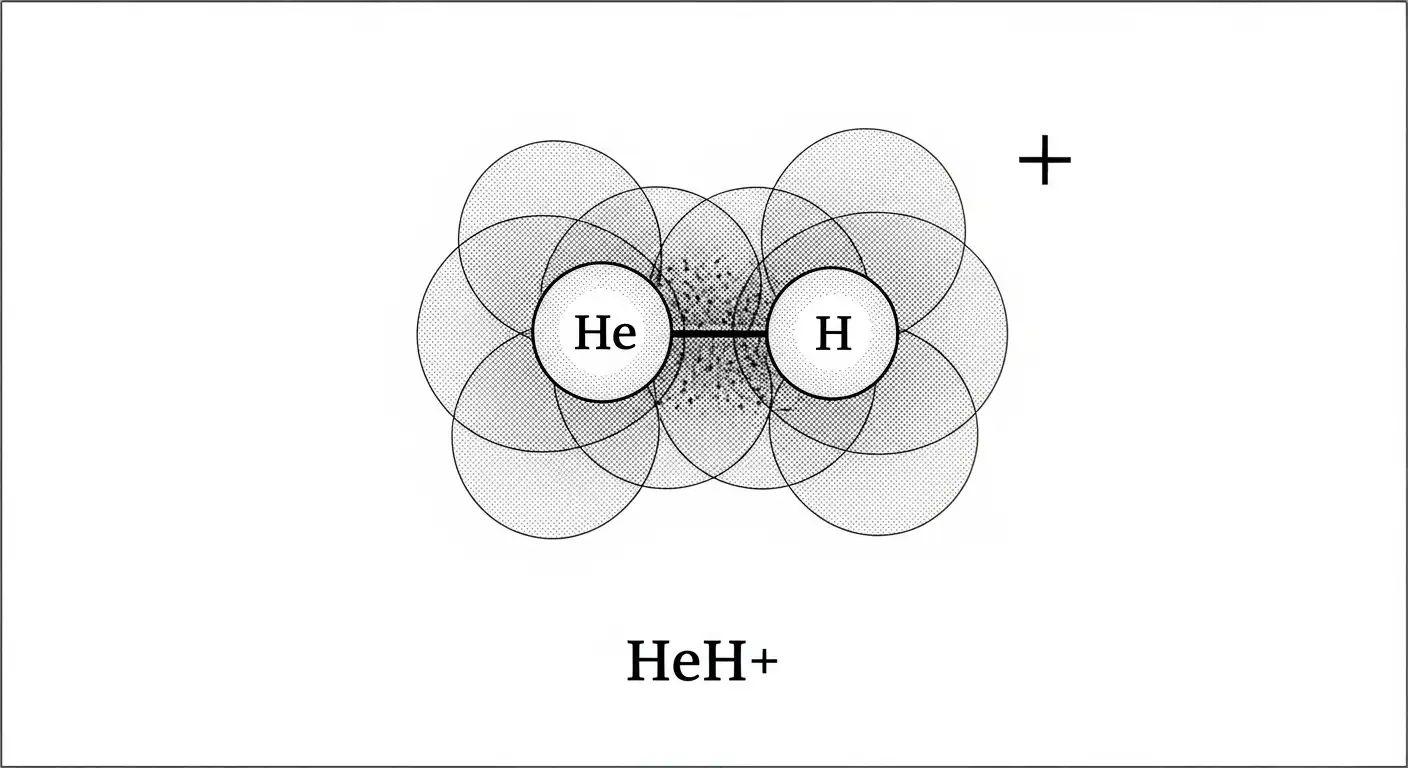
At its simplest level, Helonium (specifically the Helium Hydride ion, $HeH^{+}$) is what happens when a Helium atom meets a bare proton (a Hydrogen ion) and decides to share its electrons. In standard high school chemistry, we are taught the “Octet Rule,” which suggests Helium is perfectly happy with its two electrons and will never, ever bond with anything else.
Helonium proves that rules are meant to be broken—provided you have enough energy. It is a positively charged molecule, or a cation. It consists of one Helium atom and one Hydrogen nucleus. Because it’s an ion, it is incredibly reactive. In fact, it is widely considered the strongest known acid, because it wants to get rid of that extra proton more than almost any other substance in existence.
You won’t find a bottle of Helonium sitting on a shelf in a lab. It is a “transient” species, meaning it exists only in specific environments like the vacuum of space or inside high-tech particle accelerators. It is the ultimate chemical rebel: a bond formed by an element that is famous for not bonding.
Helonium Explained: The “Empty Seat” Scenario
To understand how Helonium works, imagine a crowded bus. The Helium atom is a person sitting comfortably in a double seat, occupying both spots with their luggage (its two electrons). They are perfectly content and have no intention of letting anyone else sit down.
Now, imagine a frantic passenger (a bare proton, or $H^{+}$) rushes onto the bus. This passenger has no seat and no luggage of their own. In the normal world of chemistry, the Helium passenger would ignore them. But in the high-energy environment of a star’s wake or a laboratory plasma, the force is so intense that the $H^{+}$ passenger essentially wedges themselves into the corner of Helium’s seat.
Helium doesn’t really want the company, but for a brief, energetic moment, they share the space. This awkward, high-tension arrangement is Helonium. It is inherently unstable because as soon as the bus slows down or another seat opens up, that proton is going to jump away as fast as possible. This is why Helonium is so reactive; it is constantly looking for a reason to stop being Helonium.
Sniffies Map App: The Ultimate Guide to Real-Time Connection
How Helonium is Created: Step-by-Step
Creating Helonium isn’t as simple as mixing two gases in a jar. It requires specific conditions where atoms are stripped of their usual defenses. Here is how the process typically unfolds in a controlled environment:
-
Ionization: You start with a chamber of pure Helium gas. Using an electrical discharge or a laser, you strip an electron away from some of the atoms or introduce free protons ($H^{+}$) into the mix.
-
Proton Bombardment: In this high-energy state, the bare protons move at incredible speeds. When a proton gets close enough to a neutral Helium atom, the electromagnetic attraction between the proton and Helium’s electrons overcomes the “nobility” of the gas.
-
The Formation of the Bond: The Helium atom shares its two electrons with the proton, creating a sigma bond. At this exact moment, $HeH^{+}$ is born.
-
Observation: Because Helonium is so short-lived, scientists use a tool called a mass spectrometer to detect its specific weight-to-charge ratio or use infrared spectroscopy to see the “vibration” of the bond before it breaks.
-
Dissociation: Usually within microseconds, the Helonium ion will collide with another molecule or a wall. Because it is the ultimate acid, it will “gift” its proton to almost anything it touches, reverting back to stable, lonely Helium.
Common Mistakes People Make About Helonium
Because this is a niche topic, there is a lot of misinformation floating around. If you want to master the concept of what is helonium, you need to avoid these three common pitfalls:
1. Thinking it’s a Neutral Gas
Many people assume Helonium is a gas like Oxygen or Nitrogen that you could fill a balloon with. It isn’t. It is an ion. Because it carries a positive charge, it is constantly seeking an electron or a way to dump its proton. You cannot have a “tank” of Helonium.
2. Confusing it with Alpha Particles
An alpha particle is just the nucleus of a Helium atom (two protons, two neutrons). While an alpha particle is involved in many nuclear reactions, Helonium is a molecule. It has a bond. Alpha particles are single pieces of an atom; Helonium is a partnership between two different elements.
3. Assuming it’s “Useless” Theory
It’s easy to think that if it only lasts for a microsecond, it doesn’t matter. However, Helonium is the “First Molecule.” Scientists believe that about 100,000 years after the Big Bang, Helonium was the very first chemical bond to form in the cooling universe. Without this weird, unstable ion, the more complex chemistry that leads to stars, planets, and humans might never have started.
Helonium vs. Helium: A Quick Comparison
To truly understand the unique nature of this substance, it helps to see how it stacks up against its parent element.
| Feature | Helium (He) | Helonium (HeH+) |
| Stability | Extremely Stable (Inert) | Extremely Unstable (Reactive) |
| Charge | Neutral (0) | Positive (+1) |
| Bond Type | No natural bonds | Single Sigma Bond |
| Acidity | Non-acidic | The strongest known Brønsted acid |
| Abundance | 2nd most common in the universe | Rare; found in nebulae and labs |
| Lifespan | Indefinite | Microseconds (in atmospheric pressure) |
Pro Tips for Understanding Exotic Chemistry
If you are studying Helonium for a chemistry course or a research project, keep these best practices in mind:
-
Focus on the Proton Affinity: Helonium has the lowest proton affinity of any neutral molecule. This is the scientific way of saying it hates holding onto its proton. Use this phrase in your papers to show a deep understanding of the material.
-
Think Spatially: Remember that Helonium is linear. Since it only involves two nuclei, its geometry is the simplest possible molecular shape—a straight line.
-
Look to the Stars: If you are looking for real-world applications, look at astrochemistry. In 2019, Helonium was finally detected in a planetary nebula (NGC 7027) using the SOFIA airborne observatory. This confirmed decades of theoretical predictions.
-
Don’t ignore the “Isotopes”: While $HeH^{+}$ is the most common form, researchers also study versions using Tritium or Deuterium to see how mass affects the bond’s vibration.
Frequently Asked Questions
Is Helonium a metal?
No, Helonium is a molecular ion. It does not exhibit metallic properties such as conductivity or luster in its standard form. It exists primarily as a plasma or a trace gas in space.
Why is Helonium called the “strongest acid”?
An acid is defined by its ability to donate a proton. Because Helium is a noble gas and “wants” to be alone, it will force its proton onto any other molecule it encounters. This makes it more acidic than even concentrated sulfuric acid.
Can you breathe Helonium?
Absolutely not. Not only is it incredibly rare and hard to produce, but its extreme acidity would instantly destroy lung tissue on a molecular level.
Where was Helonium first discovered?
It was first produced in a laboratory in 1925. However, it wasn’t until 2019 that it was actually “seen” in the wild (outer space) using infrared telescopes.
Does Helonium have a smell or color?
As an ion in a vacuum, it doesn’t have a smell. In a laboratory setting, the plasma used to create it might glow (usually a purplish-white color), but the molecule itself is colorless.
The Big Picture: Why Helonium Matters Today
Understanding what is helonium is more than just a trivia fact for chemists. It represents the “missing link” in our cosmic history. For decades, we had a gap in our timeline: we knew the Big Bang happened, and we knew stars eventually formed, but we didn’t see the bridge between the two. Helonium is that bridge. It was the first time gravity and electromagnetism conspired to create a complex structure from the chaos of the early universe.
If you are a student or a science enthusiast, let this be your takeaway: In chemistry, nothing is impossible—only difficult. Even the most “anti-social” elements like Helium can be forced into a relationship under the right circumstances.
Your next step: To see this science in action, I recommend looking up the “SOFIA telescope 2019 Helonium discovery” results. Seeing the actual spectral data from a nebula millions of light-years away makes this theoretical ion feel much more real.